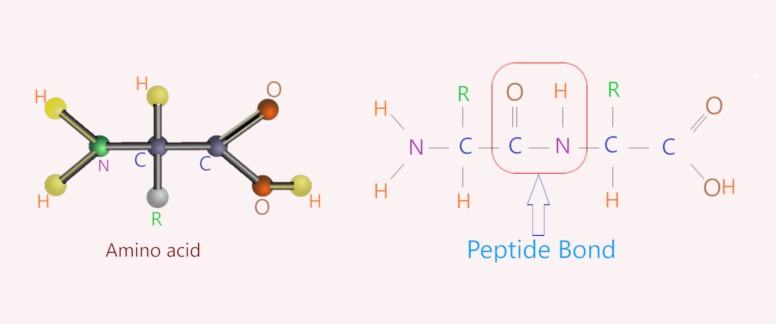
What compounds form polymeric chains?Ģ.1. Homopolymers are polymers made by joining together monomers of the same chemical composition or structure. Polymers are chains with an unspecified number of monomeric units. Polymers are a class of synthetic substances composed of multiples of simpler units called monomers. The synthetic material nylon is another example of this type of polymer. There are two basic ways to form polymers: (a) linking small molecules together, a type of addition reaction, and (b) combining two molecules (of the same or different type) with the elimination of a stable small molecule such as water. One monomer gives up a hydroxyl (OH) group and one gives up a (H). One example of a polymer is poly(ethene). Joining two monomers is achieved by a process known as dehydration synthesis. There are variable numbers of atoms in the chains of a given polymer.
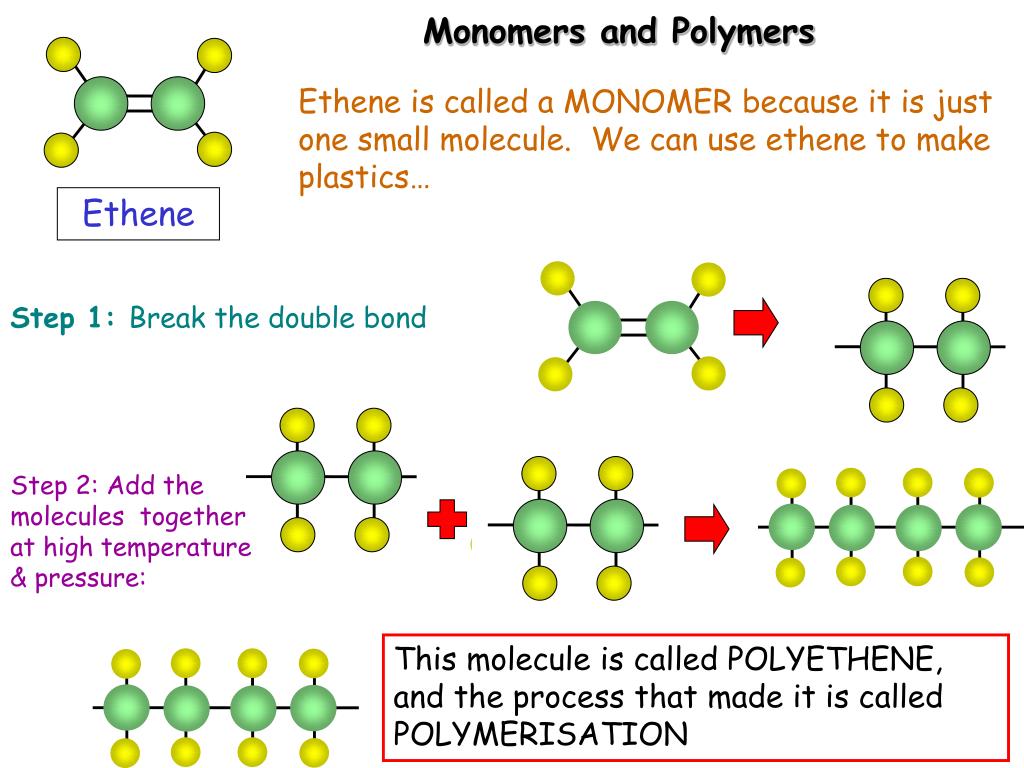

The atoms in a polymer molecule are joined together by strong covalent bonds in long chains. Polymers make up many of the materials in living organisms, including, for example, proteins, cellulose, and nucleic acids. Polymer, any of a class of natural or synthetic substances composed of very large molecules, called macromolecules, that are multiples of simpler chemical units called monomers. The monomers combine with each other via covalent bonds to form larger molecules known as polymers. Most macromolecules are made from single subunits, or building blocks, called monomers.

